Nuclear engineering is the practical application of the principles of nuclear physics and the interaction between radiation and matter. This field of engineering includes the design, analysis, development, testing, operation and maintenance of nuclear fission systems and components, specifically, nuclear reactors, nuclear power plants and/or nuclear weapons.
The field can also include the study of nuclear fusion, medical applications of radiation, heat transport, nuclear fuels technology, nuclear proliferation, and the effect of radioactive waste or radioactivity in the environment.
You may not expect to hear that the bananas you eat or rocks you walk on outside are radioactive – but it’s true! Bananas, rocks and other unsuspecting objects in our lives are actually radioactive. An object is radioactive if the small units that make up that object suddenly become unstable and begin to emit radiation, which is an energy wave moving out from an atom. It can be harmful, ranging from a sunburn caused by the sun’s radiation to very serious radiation from a nuclear power plant meltdown. Certain objects, like rocks and bananas, have very little radiation. Radioactive decay is a term used to describe the changes in an atom once it has become unstable and begins to lose its particles. In this activity, you can model radioactive decay using pennies!
Throw one hundred coins, remove all those that come up tails, place them in a pile, repeat—you’ve got yourself a hands-on model for radioactive decay. The piles graphically show the meaning of the term “half-life.”
Materials:
- 100 pennies, or you can also use M&Ms, or puzzle pieces. Anything with distinct up and down sides will work for this activity!
- 1 resealable bag
- Graph paper and data table (provided below)
- Stopwatch or timer
Instructions:
- Place the pennies in the bag and seal it.
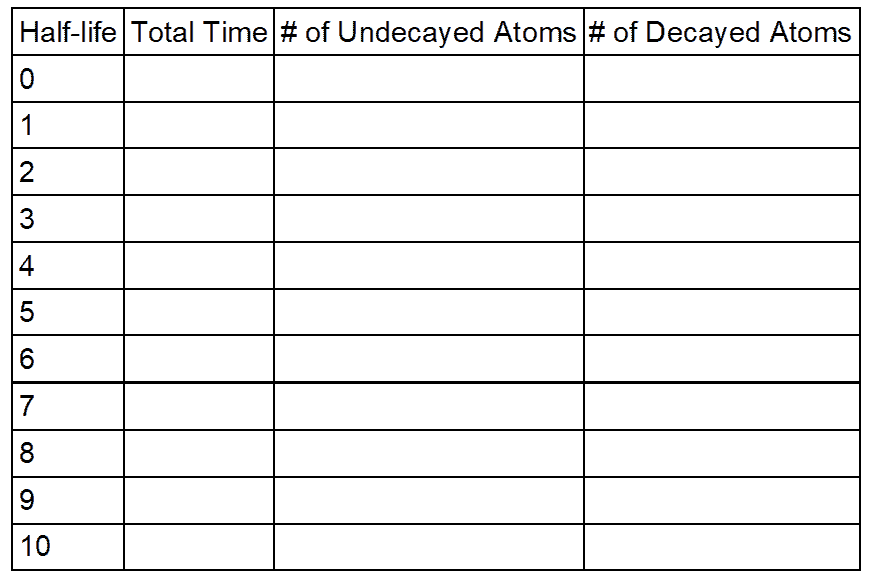
- Shake for 10 seconds. Pour out the coins and count the number that are face down, or “tails”. Record the data in the table provided. These atoms have “decayed.”
- Return only the face up coins to the bag and reseal it. Shake the sealed bag for 10 seconds and repeat the process.
- Continue shaking and counting until all the atoms have decayed.
- Graph the number of undecayed atoms vs. time.
- Repeat the experiment using half-lives of 20 seconds, and 1 minute – meaning shake the bag for that amount of time. Compare the resulting graphs.
Data and Observations:
Graph Decayed Atoms vs Time:

Author
-

SWE Blog provides up-to-date information and news about the Society and how our members are making a difference every day. You’ll find stories about SWE members, engineering, technology, and other STEM-related topics.






